 par Ulrike Marsky
par Ulrike Marsky By Dr. Ulrike Marsky, Technical Manager at Véto-pharma
Sustainable varroa control has been an ongoing challenge for beekeepers around the globe for decades.1 Although several substances are available to reduce varroa mite infestation (Varroa destructor) in honey bee colonies (Apis mellifera), all of them come along with specific requirements for use concerning factors such as ambient temperature, brood status of the honey bee colony, treatment duration, or the number of required treatment applications.
With significant variability between years in local weather, onset of flowerings, bee brood development, and varroa infestation pressure, strategic planning of the annual varroa infestation management can be difficult to anticipate for beekeepers.
One of the “side effects” of this situation is that some beekeepers prefer to rely on either a single or very few treatment options, which they perceive as particularly reliable and effective. In many cases, a synthetic miticide becomes the treatment of choice for beekeepers trying to keep their pest management strategy simple, but successful.
The problem with “keeping it simple” in the context of varroa control has been observed repeatedly in the past few decades in all global regions where varroa mites infest honey bee hives: After too many repeated applications of the same miticide in the same bee colonies, varroa mite populations become resistant against the active substance.2
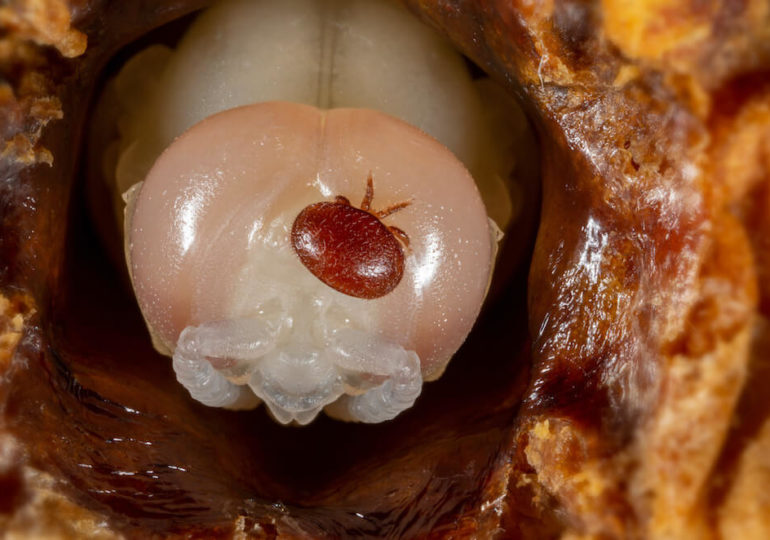
Resistance development in varroa mites has been well documented in the cases of tau-fluvalinate and flumethrin (both pyrethroids) as well as coumaphos (an organophasphate). Overall, resistance against some of the most well known synthetic miticides has developed in a time frame between just two and up to approximately 15 years of use.3-4
The formamidine amitraz represents an exception from this rule, as resistance reports in countries where amitraz is used to control varroa mites have typically occurred after longer periods of active use. In Poland, amitraz has been in use since the early 1980s, for more than 40 years, and it still demonstrates high treatment efficacy in the field.5
In France, amitraz resistance in varroa mites has been reported after a relatively long period of use (25+ years). It occurs in “patches” rather than showing the widespread and severe reduction of efficacy that has been observed for the pyrethroids and coumaphos.
The use of high-dosed off-label amitraz treatments which have been authorized for different target species (such as cattle) as well as the misuse of authorized amitraz-based varroa treatments (under- or overdosing, exceeding the recommended treatment period, etc.) represent some of the main risk factors for resistance development in varroa mites.6 The motivations for misuse or overuse of the same substance to control varroa in honey bee hives are manifold.
From lack of variety in available treatment options in some global regions to saving costs for varroa treatments to “sticking with a single treatment that works” to following recommendations of other beekeepers, a whole variety of reasons lead beekeepers to using unauthorized treatments, misuse or overuse authorized treatments.
While the selection of available authorized miticides in many global regions has increased noticeably over the last few decades, some countries still lack a sufficient variety of miticides (with different active substances) that beekeepers have legal access to. Especially in global regions with very warm climates and no natural brood break in winter, high temperatures throughout the active beekeeping season, and almost uninterrupted honey flows, a lack of available substances poses a high risk to colony health. 6-7
In addition, the availability of long-term varroa treatments as well as so-called flash treatments with a quick knockdown effect on varroa mite populations is an important element to foster a successful and sustainable treatment strategy.
By definition, a lack of available treatment options more likely leads to repeated use of the same miticide(s) over and over again, thereby favouring resistance development in varroa mites.
Global warming and other environmental changes have the potential to challenge beekeepers severely in planning their annual varroa control strategies.7
Earlier onset of spring build-up and less frequent winter brood breaks could decrease beekeepers’ opportunities for an oxalic acid treatment in the brood-free period. Instead, varroa mites would potentially reproduce throughout the year, which could impact the infestation in the new season.7-8
Other factors such as extreme droughts in spring or summer can contribute to dwindling colony health and make additional hive management actions such as supplemental feeding by the beekeeper a necessity. In these more challenging circumstances, beekeepers need to learn new strategies to reduce varroa populations in ways they may not have considered thus far.
Several organic substances (organic acids, except for oxalic acid) authorized for varroa control are distributed in the colony by temperature-dependent evaporation. The higher ambient temperatures are during the treatment period, the higher the vapour pressure during evaporation and the faster the release of the active substance in the hive. When recommended maximum ambient temperatures for these substances are exceeded, treatment applications can cause severe toxic side effects in honey bee colonies (such as queen loss or high worker bee mortality).9
Different hive management actions that either directly reduce varroa mite infestation or indirectly promote colony health represent a crucial element of honey bee health education for beekeepers. One of the most underrated hive management actions by beekeepers is the varroa mite count or varroa monitoring. The most important message for beekeepers is that monitoring must be conducted regularly, preferably with the same method, and should be well documented. Over time, monitoring enables beekeepers to detect unusual mite influx or early mite population peaks and to compare data between years.
Wax hygiene poses another example of an underrated hive management technique, although it is crucial for colony health in several ways. All miticides leave residues in honey and/or wax to varying degrees after the varroa treatment has been removed from the hive. It is therefore not recommended to apply miticides during honey flow when honey supers are set up. This is to prevent a possible contamination of honey intended for human consumption. In addition, it is indispensable for beekeepers to renew wax in their colonies on a regular basis to avoid the accumulation of pathogens, agricultural pesticides and miticides in their hive products. This is true from a sanitary perspective, but also to avoid the development of resistances: A study (by Fulton et al., 2019) demonstrated that the accumulation of tau-fluvalinate in beeswax could be transmitted to larvae and adult bees, ultimately exposing varroa mites to continuous doses of this active ingredient.10 Introducing new wax every three years or, alternatively, renewing 1/3 of the colonies’ wax combs every year is the most commonly recommended path.11
Other hive management actions are more directly linked to the reduction of varroa infestation in honey bee colonies. Drone brood removal in spring, for example by cutting out and freezing excess drone brood, can significantly delay varroa population build-up during the active season.12
Queen caging in spring or summer produces brood-free colonies, opening up the opportunity for oxalic acid treatment applications during the active season. Correctly timed, the caging of queens does not negatively affect population strength or vitality of the colony at all. Instead, it offers a welcome reduction of varroa mite pressure during the season, opening up a window for oxalic acid treatments in the absence of brood.13
Splitting colonies, producing nuclei or artificial swarms reduces the mite population in the two “daughter” colonies, compared with the initial mite load in the hive of origin. This can help prevent excessive mite infestation at the end of summer.14
All of these (and other) management actions are supportive and efficient methods to reduce mite infestation in the hive – even without applying organic or synthetic varroa treatments.


Applying a combined strategy of diagnostic (varroa monitoring), preventive (splitting colonies, producing nuclei or artificial swarms; drone brood removal; queen caging) and treatment (miticide application) actions is highly recommended to implement a sustainable treatment strategy. Instead of focusing on (pharmaceutical) varroa treatments only in their varroa control strategy, beekeepers should utilize all the different tools available to them in their goal to manage varroa infestation efficiently and sustainably.
This Integrated Pest Management (IPM) approach not only optimizes varroa control, but it also helps preventing resistance development in varroa mite populations.
Focusing more specifically on varroa treatment plans and the frequency and type of miticides applied, it is essential for beekeepers to become aware of the principle of treatment rotation. Even with other (diagnostic and preventive) varroa management actions implemented in-between miticide applications, it is still recommended to rotate active substances from one treatment to the next. If possible, beekeepers should apply the same principle here as with their overall pest management strategy and utilize all tools available in the “authorized varroa treatment tool box”.
Although several research studies have found that resistance reversal in varroa mite populations is possible when the respective substance is not applied for several years, it is the superior choice for beekeepers to prevent resistance development in the first place.
Some of the management techniques that have been mentioned in this article can be perceived by beekeepers as difficult to learn or time-consuming, but in comparison with suffering excessive colony losses as well as the loss (at least for several years) of one tool in the “authorized varroa treatment tool box”, learning how to efficiently apply these techniques and implementing them in an IPM varroa control plan in your beekeeping operation is indefinitely preferable.
1- Mondet, Fanny, et al. “Evaluation of suppressed mite reproduction (SMR) reveals potential for Varroa resistance in European honey bees (Apis mellifera L.).” Insects 11.9 (2020): 595.
2- Rasool, Kawsar, Ishtiyaq Ahad, and Rozy Rasool. “Efficacy of various botanicals and chemicals on ectoparasitic mite, Varroa destructor feeding on European honey bee, Apis mellifera.” Journal of Entomology and Zoology Studies 5.5 (2017): 589-595.
3- Milani, Norberto. “The resistance of Varroa jacobsoni Oud. to acaricides.” Apidologie 30.2-3 (1999): 229-234.
4- Elzen, Patti J., et al. “Detection of resistance in US Varroa jacobsoni Oud.(Mesostigmata: Varroidae) to the acaricide fluvalinate.” Apidologie 30.1 (1999): 13-17.
5- Semkiw, Piotr, Piotr Skubida, and Krystyna Pohorecka. “The amitraz strips efficacy in control of Varroa destructor after many years application of amitraz in apiaries.” Journal of Apicultural Science 57.1 (2013): 107-121.
6- Hansen, George. “GUIDE TO VARROA MITE CONTROLS FOR COMMERCIAL BEEKEEPING OPERATIONS.” (2021).
7- Smoliński, Szymon, Aleksandra Langowska, and Adam Glazaczow. “Raised seasonal temperatures reinforce autumn Varroa destructor infestation in honey bee colonies.” Scientific reports 11.1 (2021): 22256.
8- Nürnberger, Fabian, Stephan Härtel, and Ingolf Steffan-Dewenter. “Seasonal timing in honey bee colonies: phenology shifts affect honey stores and varroa infestation levels.” Oecologia 189 (2019): 1121-1131.
9- Hamid, Abdulkareem M. Membrane-barrier Delivery of Formic Acid Vapours to Control Varroa jacobsoni Infestation in Honey Bees Colonies. National Library of Canada= Bibliothèque nationale du Canada, Ottawa, 2000.
10- Fulton et al. An Examination of Exposure Routes of Fluvalinate to Larval and Adult Honey Bees (Apis mellifera). 2019. https://doi.org/10.1002/etc.4427
11- Al-Kahtani, Saad N., and EL-Kazafy A. Taha. «Effect of comb age on cell measurements and worker body size.» Plos one 16.12 (2021): e0260865.
12- Calderone, N. W. “Evaluation of drone brood removal for management of Varroa destructor (Acari: Varroidae) in colonies of Apis mellifera (Hymenoptera: Apidae) in the northeastern United States.” Journal of Economic Entomology 98.3 (2005): 645-650.
13- Büchler, Ralph, et al. “Summer brood interruption as integrated management strategy for effective Varroa control in Europe.” Journal of Apicultural Research 59.5 (2020): 764-773.
14- Evans, Kathleen Ciola. Evaluation of early summer splits on Varroa mite reduction and colony productivity. University of Delaware, 2015.